Some Known Questions About Philips Cpap Machines.
Saving CPAP makers in locations can likewise enhance the degradation price. This may be difficult to regulate in some places. PE-PUR audio abatement foam is a polyester-based polyurethane foam that is not dangerous when it is undamaged. If the polyurethane foam breaks down, it can produce particles and also release chemical gases. Philips CPAP Machines.
A lot more CPAP recall claims are anticipated to follow because there is evidence that Philips understood regarding the issues and also increased health and wellness dangers linked with the PE-PUR foam. In enhancement, Philip mentions they have actually been receiving complaints from users of the remembered sleep apnea makers regarding black fragments and debris in the airpath of the medical devices.
They disagree on where to hold the process. There are many sources of activity that can be taken against Philips, including: stringent product obligation as well as oversight You or an enjoyed one might be completely wounded as a result of a faulty medical tool made by Philips. Submitting a claim may pay for previous and future clinical expenses.
The smart Trick of Philips Cpap Machines That Nobody is Discussing
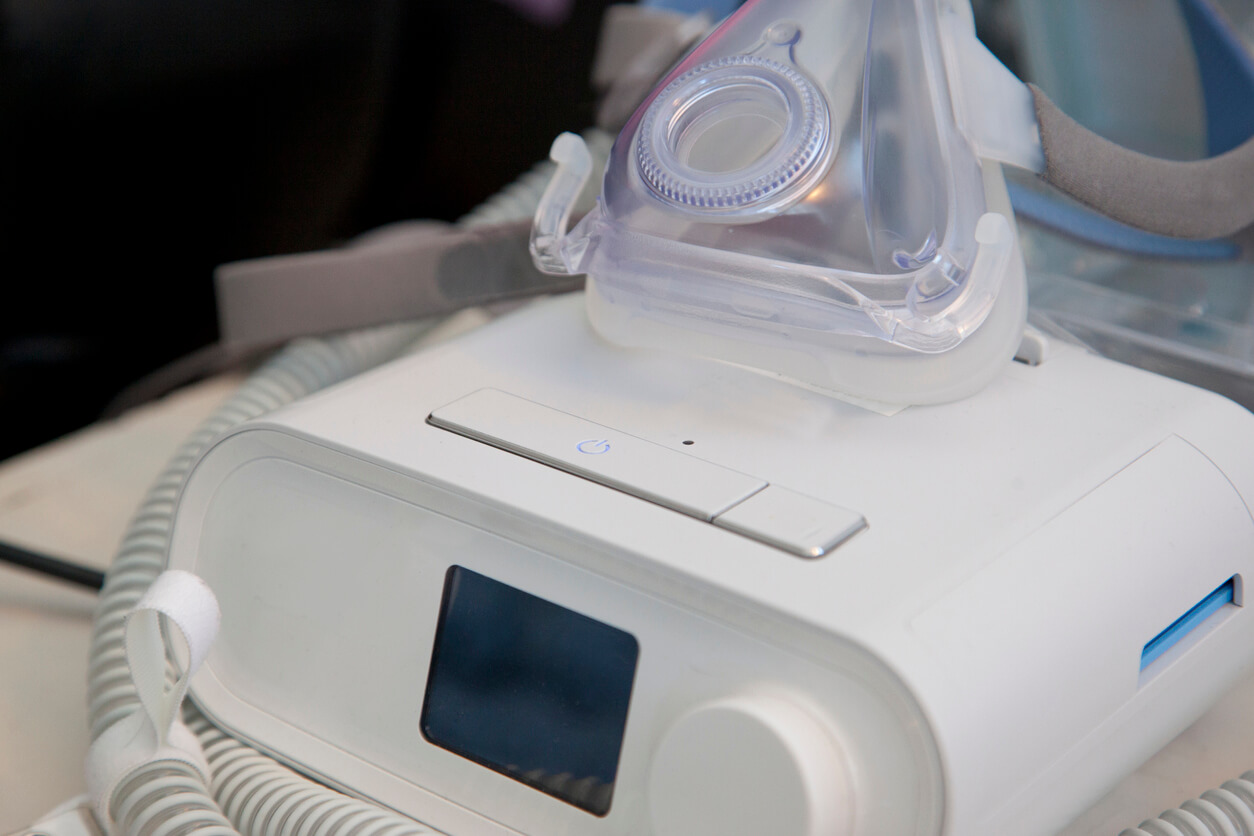
A continual positive respiratory tract stress device, even more typically described as a CPAP device, is a clinical device that is usually prescribed by doctors to deal with rest apnea conditions. There are several various classifications of sleep apnea, consisting of obstructive rest apnea, main sleep apnea, and also intricate sleep apnea disorder. Philips CPAP Machines. For those with sleep apnea or breathing conditions, CPAP as well as Bi, PAP equipments work by blowing air into patients' air passages while they are sleeping or looking for air.
Nonetheless, the Philips firm remembered its breathing equipment because of the exploration of toxins in the polyester-based polyurethane foam, which was picked for sound-reducing qualities.
The medical documents they create could assist your insurance claim later on. Defective items and medical gadgets are marketed and also remembered annually, but they are not all consulted with widescale course action lawsuits. Why is it that Philips is now confronted with class action lawsuits and also specific cases? The polyurethane foam was probably never fit to be utilized in the device's airway because it consists of hazardous chemicals.
The Facts About Philips Cpap Machines Uncovered
Purportedly, Philips had actually gotten issues regarding the foam breaking component and also being inhaled for years. Philips has been lambasted by its customers for not handling the CPAP device recall appropriately.
As an outcome of this inadequate recall, countless Americans are now battling to get any my response sleep as they go to bed each evening without an essential piece of clinical tools. There are broach adding sleep deprivation-related damages to the class activity suit, or maybe of different legal actions for these problems.
Do not deal with that obstacle alone when you might allow our highly praised accident experts to manage your instance in your place. If you have actually had a rupture operatively repaired, opportunities are excellent that the physician utilized mesh to help strengthen as well as safeguard this area. While it often works as intended, rupture mesh can in some cases fall short, causing complications.
Get This Report about Philips Cpap Machines
If it lasts months or years after rupture mesh surgical procedure, it might lead to nerve damages or chronic inflammation. The rupture mesh adheres to the intestinal tracts or relocations around in the body, blocking the digestive tract. Chronic swelling around the hernia mesh might result in infection. Implant creates problems, such a knockout post as discomfort and rupture recurrence, as well as should be gotten rid of.
Lots of problems connected to hernia repair service with medical mesh that have been reported to the FDA have actually been connected with remembered mesh items that are no more on the market. Pain, infection, reappearance, adhesion, blockage, and perforation are one of the most typical difficulties related to recalled mesh. In the FDA's analysis of clinical unfavorable occasion reports to the FDA, remembered mesh products were the main reason of digestive tract opening and obstruction issues.

3 Easy Facts About Philips Cpap Machines Described
A contingent fee agreement implies we just obtain paid if we win, which we will certainly receive our charges from the quantity paid by the Accused in the event. Please call us to discuss Click This Link the information of your case by submitting the "Demand A Free Appointment" type on this page.
In September 2021, Philips revealed it would repair or replace recalled makers because of the problematic foam. That procedure might occupy to a year, according to the firm. Philips CPAP Machines. Some individuals might choose to ask their doctor for CPAP alternatives rather. On June 28, 2022, Philips offered a research study upgrade pertaining to PE-PUR sound abatement foam testing.
Philips also claimed equipments cleansed with ozone cleansers were 14 times a lot more likely to have foam destruction. PE-PUR foam might create adverse effects since of the chemicals in the foam. Philips performed lab examinations and found at least 5 poisonous chemicals existing in foam bits and gases launched from deteriorated foam.
Comments on “The 4-Minute Rule for Philips Cpap Machines”